Importation of US-Authorized BESREMi (Ropeginterferon alfa-2b-njft) Injection Due to the Current Shortage of Canadian-Authorized PEGASYS (Peginterferon alfa-2a) Injection
Summary
See Key Messages Below
Affected products
Product Brand Name |
Dosage Form, Strength, and Packaging Format |
Country of Authorization and Identifying Code |
Manufacturer |
Importer in Canada |
|---|---|---|---|---|
BESREMi (ropeginterferon alfa-2b-njft) injection |
Injection: 500 mcg/mL solution in a single-dose prefilled syringe, for subcutaneous use (preservative free). |
United States of America |
PharmaEssentia Corporation |
Innomar Strategies Inc. on behalf of FORUS Therapeutics Inc. |
Issue
There is a critical shortage of PEGASYS (peginterferon alfa-2a) injection due to manufacturing disruptions. This shortage presents serious risks to patients in Canada who depend on interferon-based therapy for treating myeloproliferative neoplasms (MPNs). Although PEGASYS is not authorized by Health Canada for this use, it has been prescribed off-label for this condition. Given the critical medical need, Health Canada has permitted the exceptional, temporary importation and sale of a similar but not identical product, US-authorized BESREMi (ropeginterferon alfa-2b-njft), injection with English-only labels by FORUS Therapeutics Inc.
Audience
Healthcare professionals including hematologists, oncologists, hematology nurses, oncology nurses, and pharmacists.
Key messages
- There is a critical shortage of PEGASYS (peginterferon alfa-2a) injection in Canada. Given the serious risks this shortage presents to patients in Canada who have been prescribed interferon-based therapy for treating myeloproliferative neoplasms (MPNs), Health Canada has permitted the exceptional, temporary importation and sale of a similar but not identical product, US-authorized BESREMi (ropeginterferon alfa-2b-njft) injection with English-only labels by FORUS Therapeutics Inc.
- In Canada, PEGASYS is indicated for the treatment of adults with chronic hepatitis B and C. While Health Canada has not authorized its use in MPNs, it has been prescribed off-label for this condition.
- In the US, BESREMi is indicated for the treatment of polycythemia vera (PV), a type of MPN.
- Healthcare professionals are advised that:
- BESREMi contains a different active ingredient than PEGASYS and should not be considered a direct substitute.
- BESREMi has a different indication, dosing instructions, safety considerations and monitoring requirements than PEGASYS. Healthcare professionals should carefully consider these differences, and consult the US Prescribing Information (USPI) and relevant clinical practice guidelines, when evaluating treatment options.
- The US Food and Drug Administration (FDA) has approved BESREMi for safety and effectiveness, however patients should be made aware that BESREMi has not been evaluated by Health Canada for safety, efficacy, or quality. Given the critical medical need, Health Canada has permitted its exceptional, temporary importation and sale.
The BESREMi USPI, Medication Guide and Instructions For Use, available in English and French, should be consulted for information on safe use and shared with patients as appropriate.
Background
In Canada, PEGASYS is indicated for the treatment of adults with chronic hepatitis B and C. It has also been used off-label to treat MPNs.
In the US, BESREMi is indicated for the treatment of adults with polycythemia vera (PV), a type of MPN.
Information for consumers
In Canada, PEGASYS is authorized for the treatment of adults with long-term liver disease caused by the hepatitis B or C virus. It has also been used to treat myeloproliferative neoplasms (MPNs), which are blood cancers where the bone marrow makes too many blood cells.
In the US, BESREMi is authorized for the treatment of adults with polycythemia vera (PV), a type of MPN.
Due to a shortage of PEGASYS in Canada, Health Canada has temporarily allowed the importation and sale of US-authorized BESREMi. The US Food and Drug Administration (FDA) has approved BESREMi for safety and effectiveness, however patients should be made aware that BESREMi has not been evaluated by Health Canada for safety, effectiveness, or quality. Given the critical medical need, Health Canada has permitted its exceptional, temporary importation and sale. While PEGASYS and BESREMi belong to the same family of medications, BESREMi has its own dosing instructions, safety considerations and monitoring requirements.
Patients who are prescribed BESREMi should discuss any questions or concerns with their healthcare professional and follow their guidance on proper use. Regular monitoring is essential during treatment, and any side effects should be reported promptly.
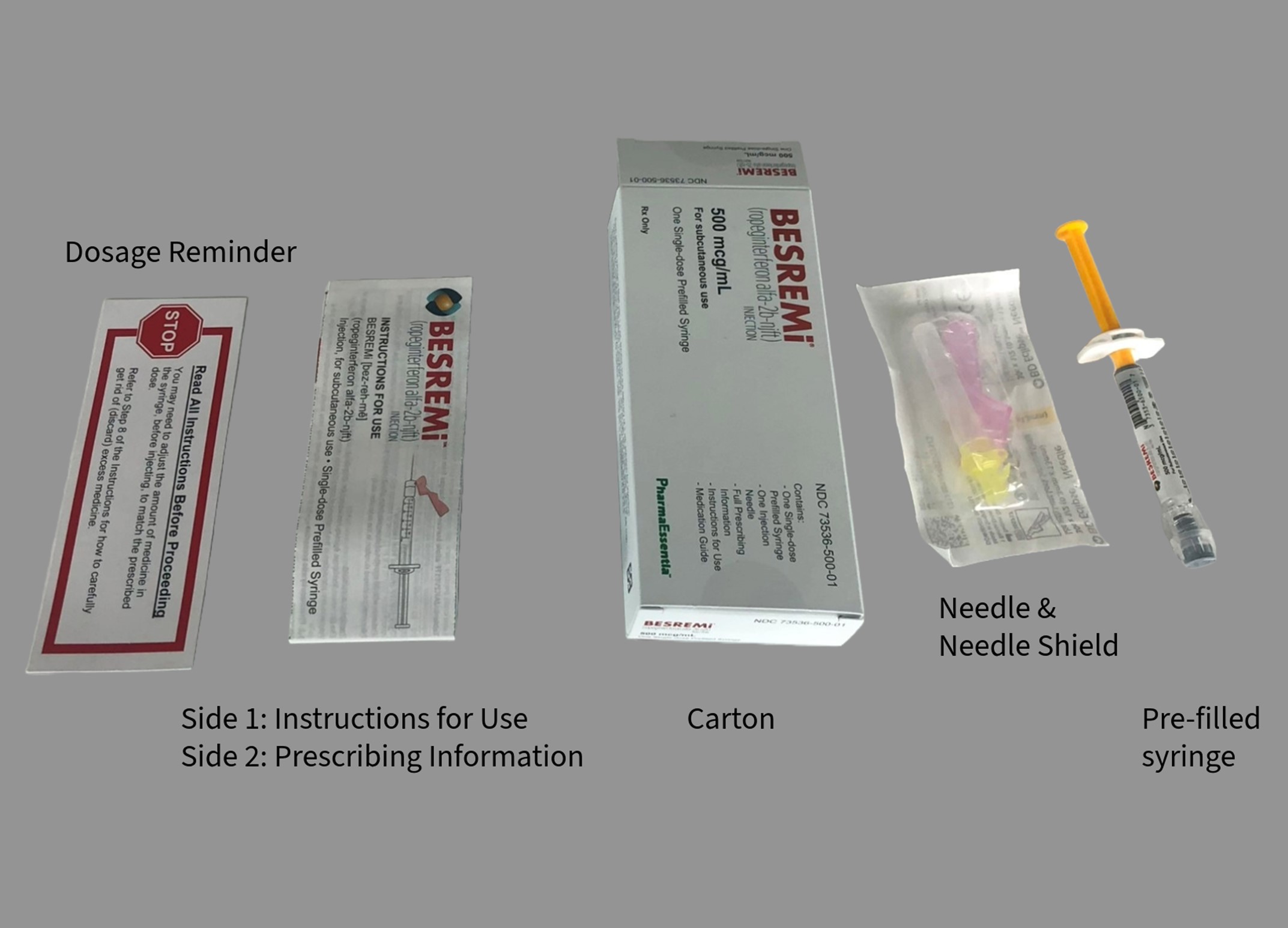
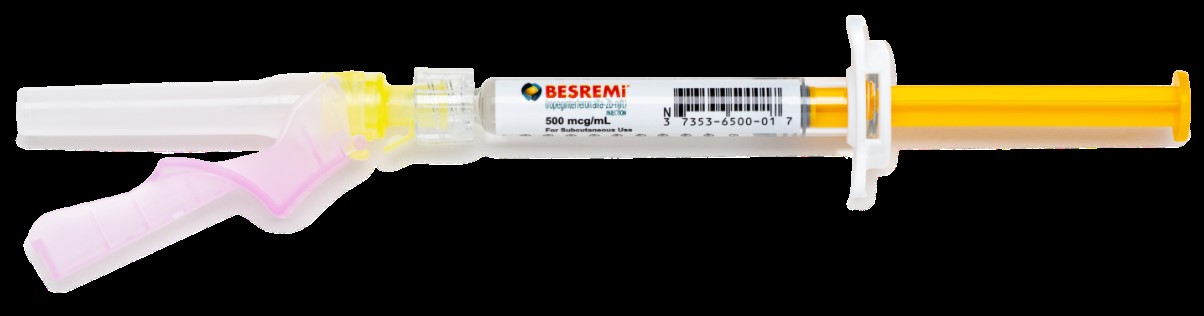
BESREMi is supplied as a pre-filled syringe containing 1 mL of solution. Patients may need to discard some of the solution in the syringe to match their prescribed dose. Discard excess medicine as per instructions.
Patients should refer to the BESREMi Medication Guide for more information, and carefully read the Instructions for Use before using the product, both of which are available in English and French.
Information for healthcare professionals
PEGASYS and BESREMi are both interferon-based therapies; however, they are distinct drugs.
Healthcare professionals are advised that:
- BESREMi contains a different active ingredient than PEGASYS and should not be considered a direct substitute.
- BESREMi has a different indication, dosing instructions, safety considerations and monitoring requirements than PEGASYS. Healthcare professionals should carefully consider these differences, and consult the USPI and relevant clinical practice guidelines, when evaluating treatment options.
- The US Food and Drug Administration (FDA) has approved BESREMi for safety and effectiveness, however patients should be made aware that BESREMi has not been evaluated by Health Canada for safety, effectiveness, or quality. Given the critical medical need, Health Canada has permitted its exceptional, temporary importation and sale.
- The BESREMi USPI, Medication Guide and Instructions For Use, available in English and French, should be consulted for information on safe use and shared with patients as appropriate. Refer to the USPI for information on use in specific populations, including during pregnancy.
Healthcare professionals are also advised to counsel patients on the points above.
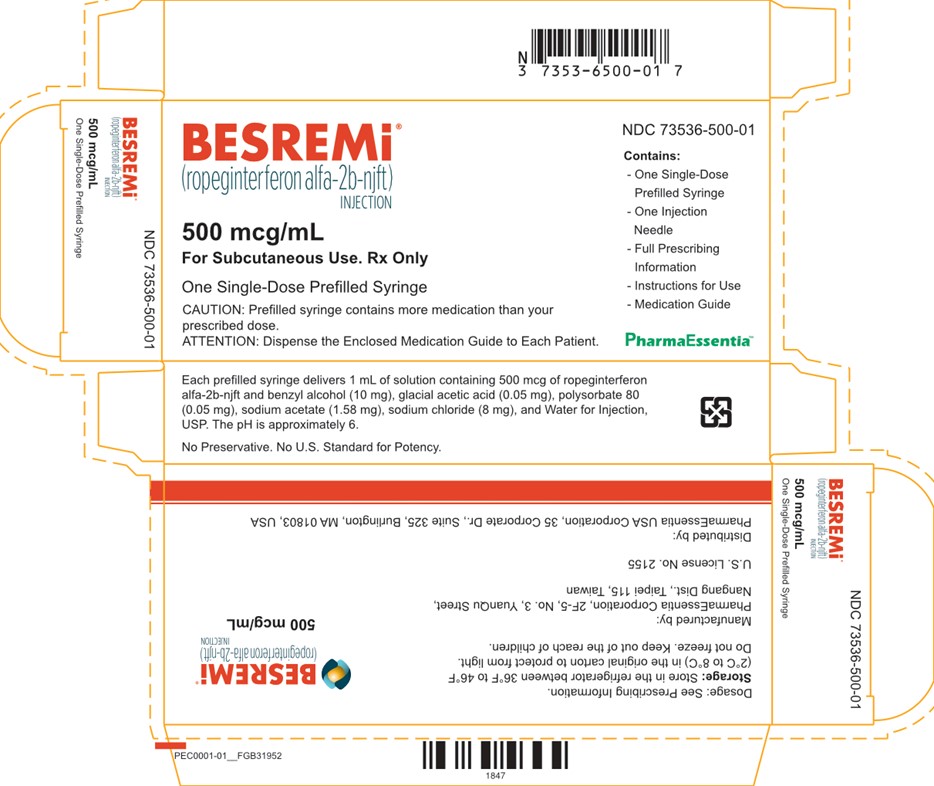
BESREMi product labels (see Appendix 1) are in English-only and do not include French text.
BESREMi will not be listed in Canadian pharmacy software systems, computerized product ordering entry systems in healthcare facilities, and health insurance system databases. A work around must be implemented by respective organizations to process and track BESREMi orders within these databases.
Additionally, BESREMi does not have a Drug Identification Number (DIN) or a barcode that scans in medication management systems in Canada. A facility-generated sticker is recommended to enable barcode scanning and allow proper identification of the product being dispensed and administered.
Action taken by Health Canada
To help mitigate the shortage of PEGASYS in Canada, Health Canada has permitted the exceptional, temporary importation and sale of US-authorized BESREMi by FORUS Therapeutics Inc. and added this product to the List of Drugs for Exceptional Importation and Sale.
Health Canada has worked with FORUS Therapeutics Inc. to prepare this alert for BESREMi. Health Canada is communicating this important safety information to healthcare professionals and Canadians via the Recalls and Safety Alerts Database on the Healthy Canadians Web Site. This communication update will be further distributed through the MedEffectTM e-Notice email notification system.
Report health or safety concerns
Managing health product-related side effects depends on healthcare professionals and consumers reporting them. Any case of serious or unexpected side effects in patients receiving BESREMi should be reported to FORUS Therapeutics Inc. or Health Canada.
FORUS Therapeutics Inc.
200 North Service Road West, Suite 129
Oakville, ON L6M 2Y1
Phone: 1-877-359-9595
Fax: 1-877-359-9590
E-mail: forus-pv@innomar-strategies.com
To correct your mailing address or fax number, contact FORUS Therapeutics Inc.
You can report any suspected adverse reactions associated with the use of health products to Health Canada by:
- Calling toll-free at 1-866-234-2345; or
- Visiting MedEffect Canada's Web page on Adverse Reaction Reporting (http://www.hc-sc.gc.ca/dhp-mps/medeff/report-declaration/index-eng.php) for information on how to report online, by mail or by fax.
For other health product inquiries related to this communication, contact Health Canada at:
Regulatory Operations and Enforcement Branch
E-mail: hpce-cpsal@hc-sc.gc.ca
Telephone: 1-800-267-9675
Original signed by

Christopher Blowes
Director, Regulatory Affairs
FORUS Therapeutics Inc., Oakville, ON
Appendix 1
A. Image of US-Authorized BESREMi (Ropeginterferon alfa-2b-njft) Injection, Carton and Carton Contents
B. Image of US-Authorized BESREMi (Ropeginterferon alfa-2b-njft) Injection, Single Use Pre-Filled Syringe with Needle Attached
C. Image of US-Authorized BESREMi (Ropeginterferon alfa-2b-njft) Injection, Syringe Label

D. Image of US-Authorized BESREMi (Ropeginterferon alfa-2b-njft) Injection, Carton Label
Additional information
Details
Get notified
Receive notifications for new and updated recalls and alerts by category.