Sunbeam® Queen Size Heated Blankets recalled due to potential fire hazard
Summary
Immediately stop using and unplug the recalled blankets and contact Star Elite for a full refund.
Affected products
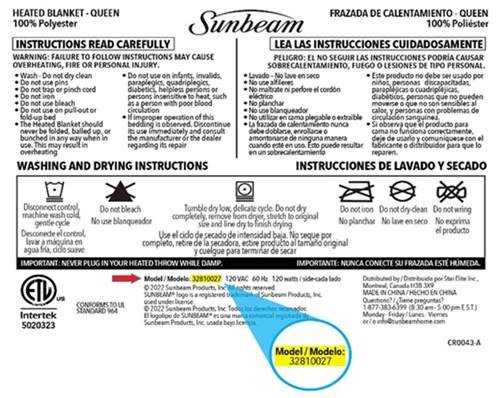
This recall involves the Sunbeam® Queen Size Heated Blanket with the model number 32810027. They were sold with detachable controllers and in the following colours: ivory, beige, light green, light blue, dark blue, burgundy, dark gray, light gray and gray violet. The Sunbeam logo and model number 32810027 are printed on the wash label affixed to the blanket.
Issue
Star Elite has learned that the Sunbeam® Queen Size Heated Blanket model number 32810027 can potentially overheat, posing possible burn or fire hazards.
As of March 8, 2023, the company has received 7 reports of incidents and no reports of injuries in Canada. In the United States, the company has received 13 reports of incidents and no reports of injuries.
What you should do
Consumers should immediately stop using and unplug the recalled blankets and contact Star Elite for a full refund. Consumers will be asked to complete the online recall registration form available at www.sunbeamhome.com/en/support/product-recall and to destroy the blanket by cutting the power cord at the plug, marking an “X” with permanent marker on the wash label and sending the recalling firm a photo of the destroyed blanket.
For more information, consumers can contact Star Elite at 877-383-6399 from 10:00 a.m.- 4:00 p.m. ET Monday through Friday or online at www.sunbeamhome.com.
Joint recall with Health Canada, the United States Consumer Product Safety Commission (US CPSC) and Star Elite.
Please note that the Canada Consumer Product Safety Act prohibits recalled products from being redistributed, sold or even given away in Canada.
Health Canada would like to remind Canadians to report any health or safety incidents related to the use of this product or any other consumer product or cosmetic by filling out the Consumer Product Incident Report Form.
This recall is also posted on the OECD Global Portal on Product Recalls website. You can visit this site for more information on other international consumer product recalls.
Additional information
Background
Number Sold
The company reported that 18,221 units of the affected product were sold in Canada and approximately 43,000 units were sold in the United States.
Time Period Sold
The affected products were sold from September 2022 to January 2023.
Place of Origin
Manufactured in China
Details
Distributor
Star Elite
Montreal, Quebec
Canada
Importer
Star Elite
Montreal, Quebec
Canada
Manufacturer
Zhejiang Lumeng Health & Technology, Co. LTD
Shoaxing City
Zhejiang Province
China
Get notified
Receive notifications for new and updated recalls and alerts by category.